  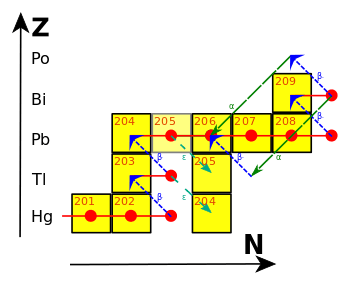 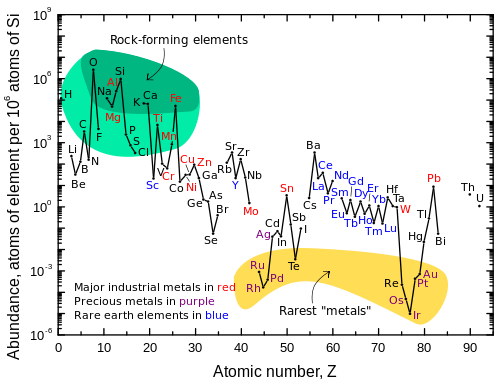
Lead (/lɛd/) is a chemical element in the carbon group with symbol Pb (from Latin:plumbum) and atomic number 82. It is a soft, malleable and heavy post-transition metal. Freshly cut, solid lead has a bluish-white color that soon tarnishes to a dull grayish color when exposed to air; the liquid metal has shiny chrome-silver luster. Lead has the highest atomic number of any non-radioactive element (two radioactive elements, namely technetium and promethium, are lighter), although the next higher element, bismuth, has one isotope with a half-life that is so long (over one billion times the estimated age of the universe) that it can be considered stable. Lead's four stable isotopes each have 82 protons, a magic number in the nuclear shell model of atomic nuclei. The isotope lead-208 also has 126 neutrons, another magic number, and is hence double magic, a property that grants it enhanced stability: lead-208 is the heaviest known stable nuclide.
| 


